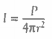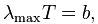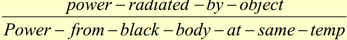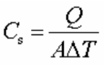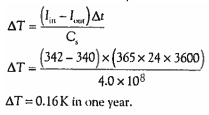Solar Radiation
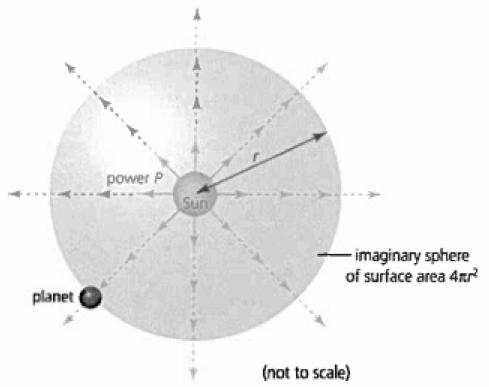
8.5.1 Calculate the intensity of the sun's radiation incident on a planet.
|
The intensity of the sun's power can be calculated at various points through the following formula:
If we imagine that the sun produces a sphere of radiation, the radius of this sphere would be the distance from the sun to the target object. The P in this case would be the luminosity of the sun (power emitted by the sun due to fusion reactions), which is defined by 3.84 × 10^26W. The radiation incident on the target object is hence represented by the intensity I.
|
The solar constant represents the average amount of incoming solar electromagnetic radiation per unit area on the earth's surface. This constant constitutes all types of solar radiation, including UV and infrared. It is thought to be approximately 1.361kW/m^2. The accuracy of the solar constant is questionable due to the following generalizations:
- This radiation is assumed to be incident on a plane perpendicular to the
earth's surface.
- It is assumed that the earth is at its mean distance from the sun.
8.5.2 Define Albedo.
Albedo is the measure of reflection on a surface. Albedo in latin refers to "whiteness" or reflected sunlight. It can be calculated by the ratio of reflected radiation from the surface to the incident radiation upon it. Being a dimensionless fraction, it can also be expressed as a percentage and is measured on a scale from zero (0%) for no reflective power to 1 (100%) for perfect reflection. An object that has no reflective power and completely absorbs radiation is also known as a black body.
The earth's albedo is 0.3, meaning, on average, 30% of the radiation incident on the earth is directly reflected or scattered back into space.
8.5.3 State factors that determine a planet's albedo.
- Seasons have different cloud formations and latitude. In thin clouds, albedo varies from 30-40% whereas in thicker clouds it can be up to 90%.
- Light surfaces such as deserts will tend to have a higher albedo while dark surfaces, such as seawater, will absorb more radiation.
- Areas of ice and snow have a high albedo compared to the rest of the planet and will reflect most of the radiation.
The Greenhouse Effect
8.5.4 Describe the greenhouse effect.
The radiation coming from the sun is approximately 50% visible light and 50% infrared, with small amounts of UV light present. When UV light enters the atmosphere from the sun, it is reflected back from the earth at a lower frequency. The light, now Infrared, is no longer able to penetrate the gaseous layer in the troposphere and becomes 'trapped'. This causes an overall increase in global temperature, affecting a number of things, including arctic permafrost, climate, animal behavior and frequency of natural disasters.
8.5.5 Identify the main greenhouse gases and their sources.
|
Carbon dioxide
Source: Consumption of fossil fuels ie. petroleum, cellular respiration, decaying matter/fossilization Effect: The most abundant manmade greenhouse gas, contributes significantly to the greenhouse effect |
Methane
Source: Agricultural land (soil fermentation, animals), burning of biomass, coal mining, gas drilling Effect: 5x more effective in causing the greenhouse effect than carbon dioxide, the oxidation of methane gives off carbon dioxide and water vapour |
|
CFC’s
Source: Aerosols, solvents, air conditioners, refrigerants Effect: Responsible for 15-20% of global warming. When CFC’s are released into the atmosphere, UV radiation breaks them down into chloride ions. The chloride ions then react with ozone and break it down to oxygen; one chloride ion is capable of destroying 100,000 ozone molecules. The thinning of the ozone layer allows more UV radiation to enter the atmosphere, which results in more InfraRed being reflected back and heating up the atmosphere. |
|
* The two major components of the atmosphere -- oxygen and nitrogen -- are so tightly bound together that they do not absorb heat and therefore do not contribute to the greenhouse effect.
8.5.6 Explain the molecular mechanisms by which greenhouse gases absorb infrared radiation.
High-frequency light (UV) is energetic and able to break bonds within molecules. Infrared light, on the other hand, causes atoms to vibrate. The greenhouse gases have a natural frequency that falls in the infrared region, so when they are hit by infrared light, they begin to resonate, creating a change in molecular dipole moment. They absorb the infrared radiation and “re-radiate” it back into the biosphere. Essentially, the greenhouse gas molecules are just the right size for the infrared radiation to resonate with the molecules, causing the molecules to heat up.
8.5.7 Analyse absorption graphs to compare the relative effects of different greenhouse gases.
In order to examine the vibrations of greenhouse gases, an infrared spectrophotometry graph is created:
The peaks in the graph to the left represent asymmetric vibrations that create a change in the molecular dipole moment of the various molecules. This state is also known as “infrared active”, while the troughs represent times when the molecules are “infrared inactive”.
8.5.8 Outline the nature of black-body radiation.
A black-body is an object that is capable of absorbing all electromagnetic radiation within its surrounding area. The black-body is at thermodynamic equilibrium; its temperature is constant. The radiation emitted by a black body has a special spectrum and intensity that depends on the temperature of the body.
A black body in thermal equilibrium has two notable properties:
A black body in thermal equilibrium has two notable properties:
- It is an ideal emitter: it emits energy that is more or equal to any other body at the same temperature, at every frequency
- It is a diffuse emitter: the energy is radiated isotropically, independent of direction
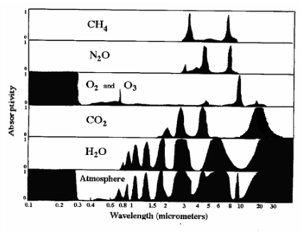
8.5.9 Draw and annotate a graph of the emission spectra of black bodies at different temperatures.
Since an object cannot emit radiation over all frequencies at once, black-body radiation is shown as the intensity distribution of the frequencies emitted. As an object gets hotter, it emits more radiation, therefore the graph shifts upwards. The area under the graph indicates the total power emitted per unit area. As temperature increases:
The peak wavelength is defined by the wavelength where the most energy is being emitted. Wien's Law describes the relationship between the temperature of the black body and its peak wavelength:
Where B is Wien's constant -- approximately 2.8977721(26) × 10^−3 mK.
8.5.10 State the Stefan-Boltzmann law and apply it to compare emission rates from different surfaces.
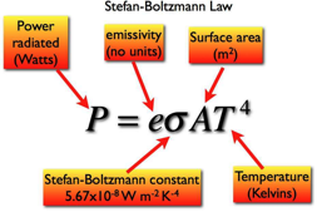
This formula calculates the amount of power radiated by an object. A black body would have an emissivity of 1, therefore the e would be omitted. If a question asks for the power radiated per unit area, the A value can be omitted.
8.5.11 Apply the concept of emissivity to compare the emission rates from different surfaces.
Since the earth is not a black body, it has a certain emissivity value. Emissivity is defined as the power radiated by a surface divided by the power radiated from a black body of the same surface area and temperature. In simpler terms, it is the relative ability of a surface to emit energy by radiation. A true black body would have an emissivity of 1 while highly polished silver could have an emissivity of around 0.02. Emissivity is a dimensionless quantity.
8.5.12 Define surface heat capacity.
Surface heat capacity is defined by the energy required to raise the temperature of a unit area of a planet's surface by one degree Kelvin. It is measured in joules per meter squared per Kelvin.
(where Q is the thermal energy transferred, A is the area and T is the temperature)
8.5.13 Solve problems on the greenhouse effect and the heating of planets using a simple energy balance climate model.
This formula is used to predict future changes in the earth's climate. Consider the following example:
In this example, a reduction of outgoing intensity by 2W per meter squared (the current average intensity is 342) would result in an increase of 0.16K. This equation inaccurately suggests that reduced activity would continue to increase global temperature. By using this equation to model climate change, we would be making the following mistakes:
- Any increase in surface temperature would also increase the outgoing radiation, according to the Stefan-Boltzmann law.
- The calculation ignores possible changes to the Earth's climate due to increasing temperatures (ie. increased cloud cover, melting ice etc.)
- The model does not consider changes in human activity
- It does not take other natural processes that periodically affect the earth's temperature into account